




Invited talk
Probing the role of vibrations in dissociative chemisorption by state resolved molecular beam/surface experiments
Surface Dynamics Group, Ecole Polytechnique Fédérale de Lausanne, 1015 Lausanne, Switzerland
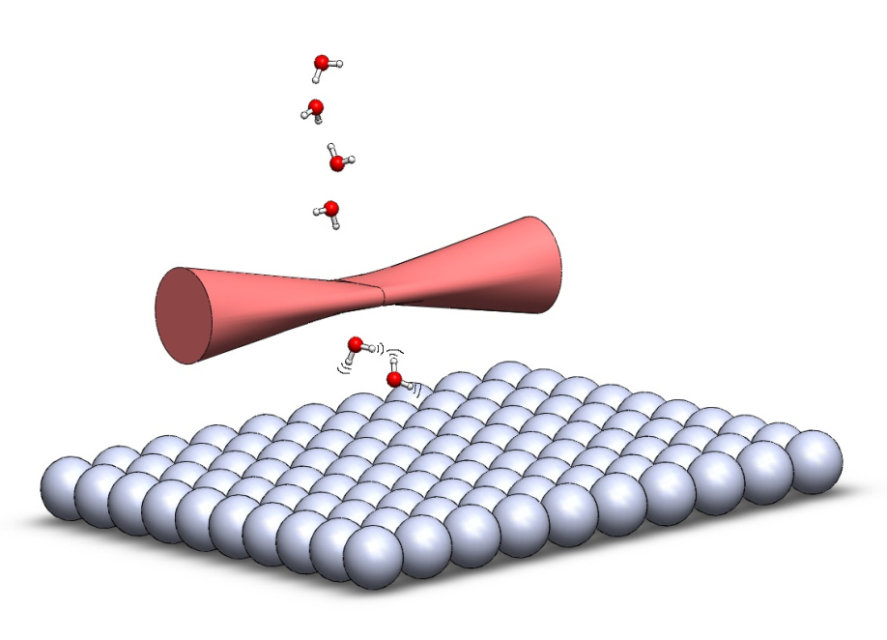
We present recent results from our laboratory on quantum state resolved reactivity measurements for the dissociative chemisorption of methane and water and their deuterated isotopologues on Ni and Pt surfaces [1-5]. Both dissociation reactions play an important role in the steam reforming process used to convert methane and water into a mixture of hydrogen and carbon monoxide by heterogeneous catalysis. Using state-selective reactant preparation by infrared pumping in a molecular beam, we prepare the surface incident methane and water molecules in specific ro-vibrational quantum states and measure the state-resolved reactivity on a single crystal surface using surface analytical techniques such Auger electron spectroscopy, King & Wells beam reflectivity, and reflection absorption infrared spectroscopy of the surface bound reaction products. We also probe the quantum state distribution of the non-reactively scattered molecules by combining infrared laser tagging with bolometric detection. The results of our measurements provide evidence for mode- and bond-specificity as well as steric effects, which shows that direct chemisorption reactions of both methane and water cannot be described by statistical rate theory but require dynamical treatments including all internal vibrational and rotational degrees of freedom of the dissociating molecule. The detailed reactivity data obtained in our measurements serves as stringent test for the development of a predictive understanding of these industrially important gas/surface using first principles theory [1,4-6].
[1] P. M. Hundt, B. Jiang, M. E. Van Reijzen, H. Guo, and R. D. Beck, Science 344, 504 (2014)
[2] P. M. Hundt, M. E. Van Reijzen, H. Ueta, and R. D. Beck, J. Phys. Chem. Lett. 5, 1963 (2014)
[3] H. J. Chadwick, P. M. Hundt, M. E. Van Reijzen, B. Yoder and R. D. Beck, J. Chem. Phys. 140, 34321 (2014)
[4] F. Nattino et al., J. Phys. Chem. Lett. 5, 1294 (2014)
[5] H. Ueta, L. Chen, R. D. Beck, I. Colon-Diaz, and B. Jackson, Phys. Chem. Chem. Phys. 15, 20526 (2013)
[6] B. Jackson and S. Nave, J. Chem. Phys. Lett. 138, 174705 (2013)